Biological Products:
Bioaugmentation products for Wastewater applications in Papermills, Refineries, Chemical, Tanneries, Municipalities, Textiles, Steel, Agriculture, Animal feedlot, Gun Powder plant, Food and Beverage- Dairy Products, Orange Juice factory, Wineries, Cookie factory, Vegetable processing plant, Meat packing, Barbecue Restaurant, Aquaculture, Ornamental Ponds for algae control, CAFO, Nursing homes, Military, Campgrounds, Universities, Regulatory agencies
Lab Services:
Filamentous Identification Lab Service. One reason to identify filaments is to determine the filaments characteristics and then determine the type present. If the type is found out, a root cause can usually be associated with a particular filament. If the cause is known, then a correction can be made to alleviate problems. Chlorination is only a quick fix. Without process changes, filaments will grow back after chlorination. Wastewater Biomass Analyses and Cooling Tower Analyses also available
Training Materials:
Training is an integral part of any job. Not everyone is at the same level of training. Many people want beginning concepts and basics. Some need technical information or troubleshooting. Some want equipment, technology or process information. We have developed a full set of Basic training, Advanced training, Filamentous Identification the Easy Way as well as custom training CD's Manuals. We also provide hands-on training classes and soon will have an Online "E-University".
Audits and Consulting:
At Environmental Leverage® Inc., we have a team of experienced individuals who come into your plant with a fresh pair of eyes. The system is checked from influent to effluent. System optimization, equipment efficiency and operational excellence are key components explored. Key Benefits Equipment efficiency Total Cost of Operation reductions Reliability and safety An onsite audit is conducted to examine system parameters, process controls, and current monitor and control procedures. A physical walk-through is conducted, process flow diagrams are examined, previous design criteria are examined and current standard operating procedures are evaluated along with data logs.
|
Evaluate Biological ProductsLatest News!
What's New!
We have just added "Virtual Audits" to our capabilities. Check out our new Services. We are in the process of developing new courses for our ""Online E-University" in order to meet the needs of our global customers that cannot travel to our public classes.Visit our new website www.WastewaterElearning.com/Elearning How do I evaluate Biological products?
It is confusing, how do they all compare?
Well one thing to note- cfu stands for colony forming units. It is a measure of plate counting bacteria to get official numeration of the amount of bacteria present per gram of product. Make sure the CFU or plate counts are per gram vs. per gallon, per lb, etc.
What you are measuring has to correlate to how you obtained the sample, how much sample is used and how the results were achieved. Sound complicated, well it can be. Sometimes vendors use plate counts as a "magic number" to wave around and try to out maneuver their competitors. Numbers are not always as important as the types of bacteria present.
We have done years of studies that prove that the number of bacteria present is not as critical as the types of bacteria present. We have found in most wastewater treatment facilities, that it is the Gram Negative bacteria that are better floc formers. Bacillus and Gram positive bacteria do not form as solid of a floc structure. Both types of bacteria will degrade BOD similarly. But in most treatment plants, BOD as well as TSS is important, therefore, floc structure is very important.
These studies use various strains of bacteria and different supplier's products to test counts vs. types of bacteria present. BOD removal, floc formation, higher life forms present, and TSS were all measurements that were taken into consideration. If plate counts are performed, the number of bacteria, the size and shape of the colonies on the plate as well as the diversity must be taken into consideration. Slime formation on the plates should be evaluated also.
*Note- Sometimes if companies are aware that a biological product is
going to be used for comparison studies between other companies and
competitive biological products, they have been known to "spike " the test
sample to a higher concentration of bacteria in order for their product to
deliver higher results. Be careful what you tell the supplier or make sure
that what sample they deliver meets those same specs continually. Hold them
to the plate counts on their biological product if they maintain that this
is their normal product quality.
Here is a photo of two sets of plates. One biological product is mostly bacillus or gram positive bacteria. The set on the right is a mixture with a high blend of Gram negative bacteria and mostly pseudomonas. The plate on the right, regardless of the initial counts, showed higher growth potential and activity, more natural polysaccharides, which help to form floc and reduce TSS and polymer consumption in final clarifiers.
Here is a two-week trial of our biological product vs. a competitor product. Our biological products typically have only a count of 1 billion per gram, but it is mostly gram negative bacteria. Here is a competitor product used at an outside lab in a treatability study. Formula A has a count of 5 billion per gram. This study was run for three weeks.
What you can see from the trial is that even though two sets of flasks were run of each biological product, a 1% solution and a 3 % solution. Formula A had significantly higher counts in the original product ( 5x109), but it could never reach the level of activity that our MicroClear formulation ( 1x109) achieved instantly. MicroClear was also able to stay at the higher level of activity throughout the experiment. Formula a peaked at day 3, dropped off at day 7 and stayed at a significantly lower level throughout the entire experiment than the MicroClear product. Our product has completely different species, not just filler bacteria that give great counts on initial product specs.
Also note, that 3% of Formula A could not reach the level of only 1% of MicroClear in spite of the higher initial counts and in spite of the 3 times the dosage in the 3% vs. 1% flask.
This proves that counts on biological products are meaningless.
Growth and activity
How do I evaluate biological products from one supplier to another? Treatability Study Comments: The treatability of the wastewater samples is determined on the basis of
Ok, well that was a lab test, sometimes field tests may prove to be different. do you still doubt that bacterial counts are not as important as types? Think of it this way, if you were to chose a basketball team and had 10 guys off the street vs. 5 Michael Jordans- who do you think would give you better results? You do not chose a polymer program based upon actives or molecular count, but on performance, why should you choose your bacterial products based upon numbers?
Still not convinced? Here is an actual papermill that was using Formula A. They were using 3-5 lbs of Formula A per day. We switched them to a program with 1 lb of our MicroSolv 118 and one lb of MicroClear M100- a micronutrient formulation. Lower dosages, yet better results, you can't beat that!
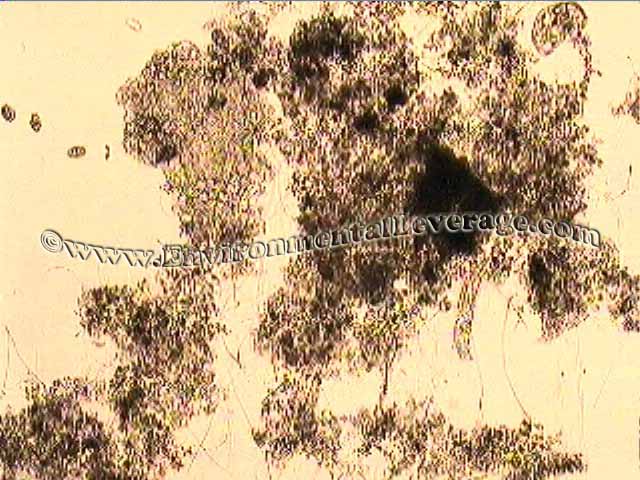
Papermills: Case History with Total System Optimization: 100% Recycle papermill with once through lagoon The first stage of the lagoon was aerated, second and third settling lagoons. Large amounts of algae, scum and duckweed covered the last two stages of the pond. This pond was on a bioaugmentation program for years.
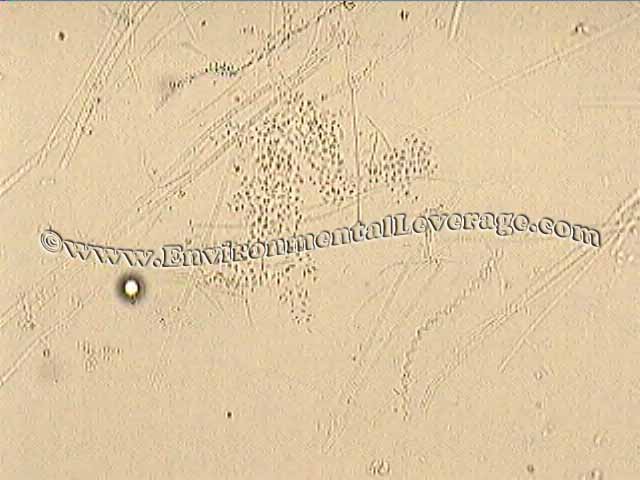
Smaller floc with lots of filaments Zooglea and spirillum Photo taken at 100x bright field Photo taken at 400x bright field
An audit was conducted, recommendations to move one of the aerators to the first half of the second stage were made to allow more oxygen to carry through the rest of the system. Bioaugmentation was changed from 2-5 lbs of product per day from local supplier to 1-2 lbs of MicroSolv 118 and 1 lb of Micronutrients M100.
In less than 2 weeks, the scum was gone off the pond, the BOD and TSS removal improved, floc structures increased significantly and higher life activity went sky high. Short, free-floating filaments disappeared. Spirillum (usually an indication of septic conditions) and zooglea were gone after changes in treatment. Some filaments are still in the floc structures, mostly Type 021N, but that is due to solids handling problems in the primary clarifier that are under consideration for optimization.
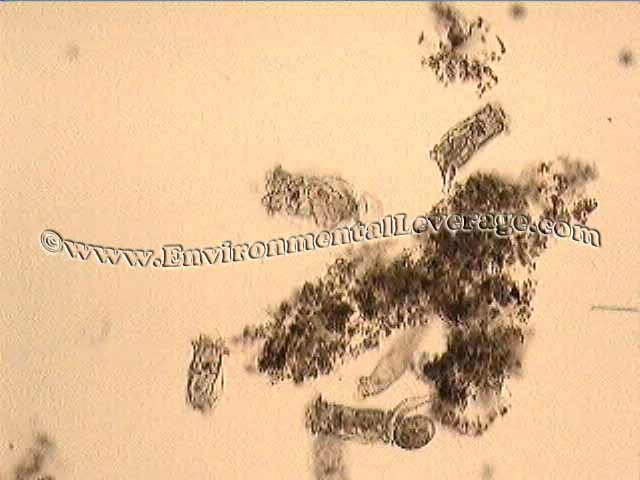
After addition of MicroSolv 118 and Micronutrients MicroClear M100
Large compact floc structures
Significant increase in higher life forms, rotifers, less filaments and TSS
The primary clarifier still needs a bit of solids handling optimization, but the plant is waiting on an additional tank up front to store settled solids that are recycled back into the system. The plant at one point had an upset for a two-week period; twice the loading of BOD was entering the lagoons. The final BOD was still below previous year's final effluent values and way below permit levels!
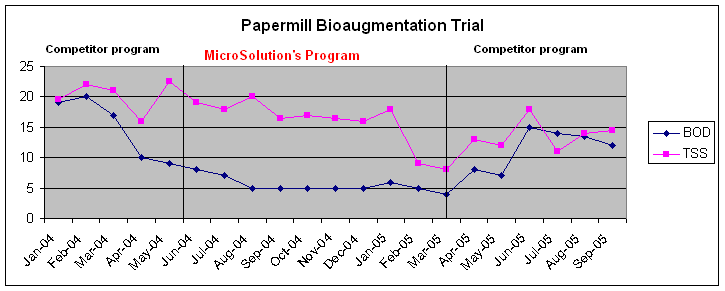
Here is the current update on our Papermill! We were asked over a year ago to come in and troubleshoot for a papermill with a once through lagoon. We made some process recommendation, gave some training and revised their current bioaugmentation program. The final effluent had the best BOD in the history of the plant. But, since the plant was on a "sole source " agreement with a major supplier of all their chemicals, they were forced to go back to their original supplier for biological products. Well, after just 6 months, the plants is back on our products and program. The results could not even come close on their program!
While we believe some of our products are specially formulated and are very good, the major thing we believe is important is the training, consulting, analyses and support that we have to offer. But the real proof is in the final BOD number! Check to see how we compare. . . . BOD final permit limits are 25 and TSS numbers are 30 ppm. We consistently hit close to 5 ppm BOD once the program was up and running! Very good for a papermill! Even the cold weather months with a normal slowdown in activity, the BOD was still on the mark! TSS is more a function of the primaries and they just installed a number of new thickeners, a new sludge recycle pump during various points in this trial, so TSS was not really an issue between both companies unless you were to say the influent loading was higher, but BOD was the real driving issue!
Call today to find out more information on how to start your own Bioaugmentation program! Additional Comments: If using this for a guideline to compare biological programs and vendors, please take into consideration Total Program value, cost per equivalent product in ratio, evaluations of shake flask testing, BOD/TOC as well as TSS comparisons. Health of the biomass after addition of product, Technical support, training, program consulting, experience and additional corporate back-up are also considerations that need to be accounted for. Many times the EPA can be involved with plants, new permit evaluations, etc. make sure your vendor is capable of providing you these services and recommendations if needed.
Products for use in Wastewater Applications for Bioengineering and BioaugmentationList of Bioaugmentation products and Links to product bulletins
Links to Additional Troubleshooting Pages For More information on Products:Biological Products, Descriptions, Usage and Applications Case HistoriesBiological Products for use in Wastewater Applications for Bioengineering and Bioaugmentation
Biological Wastewater Treatment Products
How are your bugs doing?
|

 It
is very confusing, one person says his biological product has 1 billion
count in the final product, Another vendor says his biological product is
better because it has 5 x 109 cfu, still a third says
his product has "3+ billion per gram viable cell count". What is the
difference and what do those terms mean?
It
is very confusing, one person says his biological product has 1 billion
count in the final product, Another vendor says his biological product is
better because it has 5 x 109 cfu, still a third says
his product has "3+ billion per gram viable cell count". What is the
difference and what do those terms mean?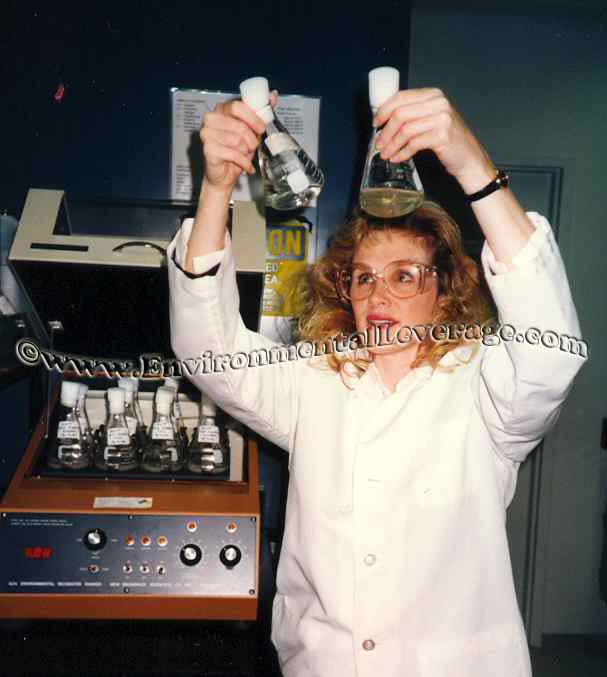 One
thing we did when testing and developing biological products was to perform
shake flask tests. Shake flask studies are conducted for treatability
studies of wastewater samples to reduce overall BOD or to target a specific
compound and to improve settling characteristics. It can be used for
toxicity studies or for product screening procedures. It is similar to a
quick and dirty jar testing for polymers that most people are familiar with
but takes a little bit more time and is used to evaluate the performance of
biological products.
One
thing we did when testing and developing biological products was to perform
shake flask tests. Shake flask studies are conducted for treatability
studies of wastewater samples to reduce overall BOD or to target a specific
compound and to improve settling characteristics. It can be used for
toxicity studies or for product screening procedures. It is similar to a
quick and dirty jar testing for polymers that most people are familiar with
but takes a little bit more time and is used to evaluate the performance of
biological products. 
 due
to types of strains present in the biological product are really what you
should be interested in!!!
due
to types of strains present in the biological product are really what you
should be interested in!!!